Erb’s Palsy and Related Brachial Plexus Birth Injuries
Families of children with injuries to the brachial plexus at birth are often traumatized by a difficult delivery. The moments after delivery are chaotic and families are unsure of what has happened to their baby. Most families are told that the baby will recover and that they need not worry. However, about a third of babies will have persistent limitations in the injured arm into adulthood.
Finding a qualified specialist can be difficult and confusing, with many centers claiming to treat children with brachial plexus injuries having no trained specialist to manage the brachial plexus. Hospital for Special Surgery has been a destination for many types of pediatric nerve injuries that affect the upper and lower extremities, including those of the brachial plexus. Our team is made up of internationally recognized experts in nerve and specifically brachial plexus injury management.
What is Erb’s palsy (brachial plexus birth injury)?
Injuries to the brachial plexus at birth (sometimes referred to as brachial plexus palsy, neonatal palsy or obstetrical palsy) have been typically given the name of “Erb’s palsy,” because it is the most common type. Plexus Nexus, the leading international consortium of doctors and therapists who treat brachial plexus birth injuries, has suggested that “brachial plexus birth injury” (BPBI) is the preferred term to describe the injury, since not all brachial plexus injuries at birth result in an Erb’s palsy. Erb’s palsy refers to an injury to only the upper nerve roots.
BPBIs can be concerning, since nearly all of the nerve fibers that provide function and sensation to the upper limb (arm, forearm, and hand) pass through the brachial plexus. The brachial plexus is made up of 350,000 individual nerve fibers (axons) that exit the spinal cord through five openings in the spinal column to form five nerve roots. Four of the nerve roots emerge from the cervical spine (neck) and one from the thoracic spine (chest). These roots are named after the numbered cervical and thoracic spinal vertebrae from which the exit the spine C5, C6, C7, C8, and T1 (Figure 1). The nerve roots combine to form three trunks. These are the:
- upper trunk (C5, C6)
- middle trunk (C7)
- lower trunk (C8, T1).
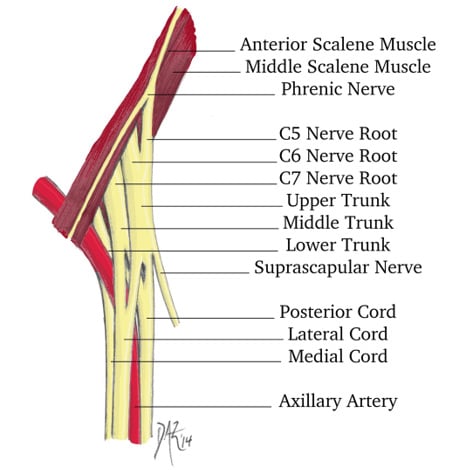
Figure 1: The Brachial Plexus (Courtesy of Dan A. Zlotolow, MD)
Although there is a lot of functional overlap, each trunk controls specific functions in the arm. The upper trunk primarily controls the shoulder and elbow bending. The middle trunk straightens the elbow, the wrist, and the fingers. The lower trunk bends the fingers and the thumb and provides fine motor control of the hand. When the brachial plexus is injured at birth, one or more of the trunks will be stretched or torn. The more roots that are involved, the less function the child will have in the upper extremity, and the more likely they are to need surgery to reconstruct the brachial plexus.
What causes a brachial plexus birth injury?
Most BPBIs occur when the baby’s shoulder gets stuck behind the mother’s pelvis during delivery, a situation called shoulder dystocia. The biggest single risk factor for shoulder dystocia is a previous delivery complicated by shoulder dystocia. The current recommendation is to perform a C-section if there was a shoulder dystocia in a previous delivery. Other common risk factors for shoulder dystocia are macrosomia (babies larger than nine pounds) and maternal diabetes. Obstetricians monitor the size of the baby with ultrasounds before the baby is born, but these can be up to 20% inaccurate. Blood sugar of the mother should also be monitored during pregnancy.
Once shoulder dystocia occurs, there are several maneuvers that can be used to try to free the shoulders, including the “McRoberts maneuver,” the “Woods corkscrew,” suprapubic pressure, delivery of posterior arm, and, in some cases, even an intentional breaking of the baby’s collar bone. If these maneuvers do not free the shoulder, there is a limited amount of time that the baby can remain stuck before they suffer from lack of oxygen, or ischemia, to the brain. Ischemia of the brain and the child can lead to cerebral palsy or even death. If the child is in severe distress, delivering a child urgently with a brachial plexus injury may be better than the alternative, and this is a judgment that the obstetrician, midwife, or other provider may be forced to make.
There are also suggestions of brachial plexus injuries occurring from muscular contraction of the uterus, or even from a rapid delivery without shoulder dystocia, but these causes are much less common. There is some evidence that the use of oxytocin (Pitocin) to induce labor or to strengthen uterine contractions may increase the risk of a plexus injury.
There are other injuries that can look like brachial plexus injuries. Fractures of the bones of the arm can cause the child not to move their arm, a condition known as a pseudopalsy, or false palsy. When the fracture heals in about 2 to 3 weeks, the arm will start moving again. Tumors of the spinal cord or infections can also cause transient or permanent paralysis.
What are the signs and symptoms of a brachial plexus birth injury?
Symptoms may include:
- Weakened or immobile upper arm (shoulder and/or elbow)
- Weak hand grip
- A limp arm
What are the different types of brachial plexus birth injuries?
The three basic types of brachial plexus birth injuries, in order of severity, are:
- Upper (or Erb’s palsy/C5-6)
- Extended upper (C5-C7)
- Global (or complete/C5-T1)
Most upper and extended upper injuries recover on their own with time. Global injuries, where the entire limb is limp at birth, usually require some form of surgical treatment.
The amount of force (stress) that each nerve root is subjected to at the time of injury leads to a stretch (strain) of the nerve root. If the strain is minimal, the nerve root will shut down for anywhere from a few hours to a couple of months and fully recover. If the strain is more severe, a majority of the nerve fibers within the nerve root will tear, while most of the structure of the nerve itself remains intact. These injuries have the potential for recovery, but because the nerve fibers (axons) have to regrow from the site of injury at about an inch a month, it takes about 5 to 6 months for recovery of functions like elbow bending and shoulder abduction, and about 18 months to reach the hand. If most or all of the structure of the nerve is torn, recovery can take up to three years to gain even elbow bending and is less certain.
Because the upper cervical roots are higher up on the neck, they tend to be injured first when the shoulder is forced downward during delivery. Think of opening a zipper with the upper roots at the top and the lower roots at the bottom. The upper roots therefore experience the most strain in global injuries where all of the roots are injured. However, the upper cervical roots have specialized ligaments that resist them from being pulled out of the spinal cord, an injury known as an avulsion. The lower roots, from C7-T1, do not have these ligaments and are therefore more at risk for avulsion from the spinal cord than the C5 and C6 nerve roots. Avulsion injuries are not recoverable, and they cannot be repaired. Therefore, even though the upper trunk sees more stress during a global injury, the lower roots may be more permanently injured.
Avulsions of the upper trunk are associated with loss of diaphragm function, leading to difficulty for the child to generate enough suction to breastfeed. Injuries of the lower trunk are usually associated with a Horner’s syndrome (ptosis, myosis, anhidrosis). Children with a Horner’s syndrome will have a droopy eyelid and a small pupil on the injured side.
How common is brachial plexus birth injury?
Brachial plexus birth injuries occur in 1 to 3 out of every 1000 births. This makes it one of the most common injuries sustained at birth. Countries with a high C-section rate or a coordinated shoulder dystocia training program tend to have a lower rate of BPBI. The rate of BPBI in the United States is among the highest in the industrialized world.
How is brachial plexus birth injury diagnosed?
The diagnosis of BPBI is a clinical one, meaning there are no reliable tests that can determine the type or severity of injury. Some centers use electrodiagnostic nerve tests such as an electromyograph (EMG) to test the condition of the muscles and the nerves, but these tests have been shown to be unreliable in babies, often overestimating the likelihood of recovery. These tests are painful, may require anesthesia, are costly, and offer no advantage over just a careful examination. If your doctor has recommended an EMG on your baby, consider getting another opinion.
Magnetic resonance imaging (MRI) has been recommended by some experts for specific indications. Imaging is not required to make the diagnosis, and it is unreliable in babies in determining prognosis or the need for surgical treatment. It also requires general anesthesia (sedation). We reserve MRI as a presurgical tool in patients with global injuries to assess the likelihood of root avulsions and, therefore, assist with surgical planning. If surgery is not indicated, an MRI is not necessary. Again, if your doctor insists on an MRI of the brachial plexus before two months of age without a surgical plan, consider getting a second opinion.
To diagnose shoulder problems in children with BPBI, the current recommendation is to use ultrasound until the child is one-year old. After that, the ultrasound becomes less reliable, and an MRI becomes more appropriate.
What happens if the brachial plexus birth injury does not recover on its own?
Most Erb’s (upper trunk) palsies will resolve on their own and do not require nerve reconstructive surgery. About half of all extended upper injuries (upper and middle trunks) will also recover. However, the majority of children with a completely limp arm at birth (global palsy) will need some form of nerve reconstruction.
In children with complete or global injuries where the arm and hand have little to no motion by three months of age, the international consensus is to reconstruct the brachial plexus with a combination of nerve transfers and nerve grafts. Nerve grafts are pieces of nerve usually taken from the leg to bridge a gap where the brachial plexus has torn. Nerve transfers are working nerves that are transferred to non-working nerves to bypass an injured area. The more avulsions there are, the more the reconstruction will rely on nerve transfers, because in an avulsion injury the root cannot be reconnected to the spinal cord, even with a nerve graft.
If the child can bend the fingers to make a fist by three months of age, most experts will wait until the child is 5 to 6 months old to see if elbow bending and shoulder function recover on their own. If not, then a reconstruction may be recommended.
All nerve reconstructions take time to recover, and recovery is never complete. Usually elbow bending returns 6 to 12 months after surgery, shoulder function improves by 12 to 18 months, and hand function begins to recover at around 18 to 24 months.
How is brachial plexus birth injury treated?
The treatment of BPBI depends on the type and severity of the injury. About two thirds of children will recover spontaneously and never need surgery. The only lasting effect may be some tightness or limited motion in the affected shoulder. This is why the first line of treatment for children with BPBI is physical therapy to mobilize and stretch the shoulder combined with braces to keep the shoulder in external rotation.
At HSS, we use the teapot splint (Figure 2) or teapot brace starting at 2 to 4 weeks of age to keep the shoulder stretched out at night. Since implementing the teapot splint, we have seen the need for surgery of the shoulder nearly go away. This has saved many children from needing tendon transfers or joint releases.
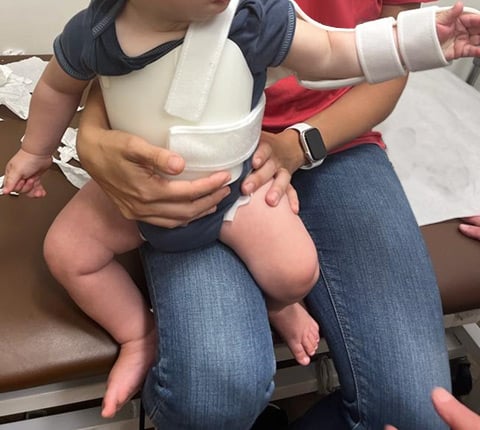
Figure 2: Teapot splint on a one-year-old child.
What happens to the shoulder if the brachial plexus birth injury is not treated?
If the shoulder is left untreated, it can deform as it grows, a condition known as glenohumeral dysplasia (Figure 3a). Glenohumeral dysplasia can get worse with age but can be severe even in children as young as three months old. Some children may dislocate the shoulder joint completely.
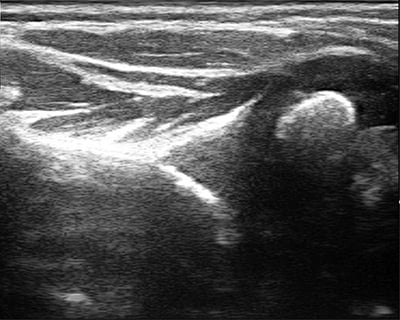
Figure 3a: Severe glenohumeral dysplasia on ultrasound
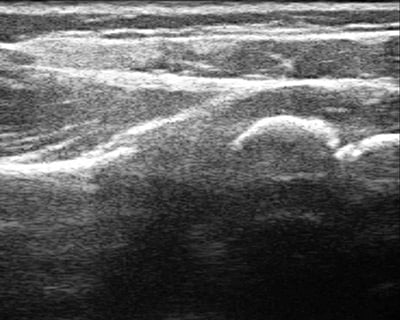
Figure 3b: Normal shoulder ultrasound
Many patients come to us too late to implement the teapot splint, and shoulder tightness with internal rotation has already set in. In children less than a year of age, it may be possible to stretch out the shoulder using botulinum toxin to temporarily weaken the muscles that turn the shoulder in, and then to cast the shoulder in a position of external rotation (Figure 4). This has to be done with the child asleep and so requires general anesthesia. Ultrasound is used to confirm that the joint is in place after the shoulder is stretched, and is it also used intermittently after the cast is removed to make sure that the shoulder is growing and developing well. A teapot splint may be used after casting for up to two years during sleep to maintain the shoulder position.
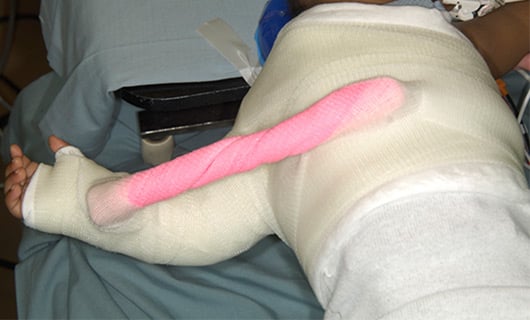
Figure 4: Shoulder spica cast placed after injecting botulinum toxin into the internal rotator muscles
If the glenohumeral dysplasia persists after nonsurgical treatment, the child may need a shoulder release with or without tendon transfers. Many different shoulder releases have been described, including an arthroscopic release. We have stopped doing arthroscopic releases in favor of an open anterior release with coracoidectomy because the results are better with lower risks in most cases.
If surgery for the shoulder is being considered in a child older than one year of age, an MRI of the shoulder is often obtained to assess the status of the joint in detail. The MRI can reveal whether or not the muscles of the shoulder are in good condition and therefore have recovered their nerve supply. The MRI can also show the shape of the shoulder joint in detail. X-rays and Computed Tomography (CT or CAT) scans are not indicated because they cannot visualize cartilage, and the bones of the shoulder joint are primarily cartilage before the age of four.
After the age of four, the shape of the glenohumeral (shoulder) joint is unlikely to change. Tendon transfers and releases are not usually performed in older children unless the shape of the joint is normal or near normal. In older children with deformed shoulder joints, options include a shape and function of the shoulder, and/or a humeral rotational osteotomy to position the arm in more external rotation.
Adults living with brachial plexus birth injuries
Many adults living with the sequelae of BPBI did not get any care for their injury during childhood. They typically suffer from limited shoulder motion, most often with internal rotation contractures of the shoulder. Their hands are always pointing towards their belly. This position of the arm is not only functionally limiting, but can cause social and self-esteem distress. As one of the few centers in the country with expertise caring for BPBI from infancy into adulthood, we have pioneered reconstructive procedures to optimize the function and appearance of the arm. Our understanding of the brachial plexus has grown exponentially in the last decade. We recommend that all adults living with BPBI seek advice from a qualified brachial plexus expert familiar with the care of adults and children.
Are there any resources for support or to learn more about BPBI?
Our plexus experts helped to create Plexus Nexus, the North American Brachial Plexus Accreditation Board, and Little Arms.org all of which are focused on improving the care and understanding of BPBI. The United Brachial Plexus Network is a support group that meets every other year in support of children and adults with injuries to the brachial plexus. Our experts have been featured at their meetings and webinars.
Explore Related Patient Stories
View All Patient StoriesReferences
- Berger A, Zlotolow DA. Brachial Plexus Birth injuries. Semin Plast Surg. 2023 Jul 26;37(2):84. doi: 10.1055/s-0043-1768686. PMID: 37503526; PMCID: PMC10371398. https://pubmed.ncbi.nlm.nih.gov/37503526/
- Kluemper C, Aversano M, Kozin S, Zlotolow DA. Long Thoracic Nerve Transfer for Children With Brachial Plexus Injuries. J Pediatr Orthop. 2021 Jul 1;41(6):374-378. doi: 10.1097/BPO.0000000000001774. PMID: 34096554. https://pubmed.ncbi.nlm.nih.gov/34096554/
- Koshinski JL, Russo SA, Zlotolow DA. Brachial Plexus Birth Injury: A Review of Neurology Literature Assessing Variability and Current Recommendations. Pediatr Neurol. 2022 Nov;136:35-42. doi: 10.1016/j.pediatrneurol.2022.07.009. Epub 2022 Jul 20. PMID: 36084421.
https://pubmed.ncbi.nlm.nih.gov/36084421/ - Louden E, Marcotte M, Mehlman C, Lippert W, Huang B, Paulson A. Risk Factors for Brachial Plexus Birth Injury. Children (Basel). 2018 Mar 29;5(4):46. doi: 10.3390/children5040046. PMID: 29596309; PMCID: PMC5920392. https://pubmed.ncbi.nlm.nih.gov/29596309/
- Russo SA, Killelea CM, Zlotolow DA, Kozin SH, Rodriguez LM, Chafetz RS, Richards JG. Scapular Stabilization Limits Glenohumeral Stretching in Children With Brachial Plexus Injuries. J Hand Surg Am. 2019 Jan;44(1):63.e1-63.e9. doi: 10.1016/j.jhsa.2018.04.025. Epub 2018 Jun 20. PMID: 29934088. https://pubmed.ncbi.nlm.nih.gov/29934088/




