Laboratory of Cartilage and Meniscus Mechanics
Our goal is to understand the relationship between joint mechanics and the biological response of tissues.
We use this information to:
- understand the effect of injury and repair on the mechanical and biological response of the knee joint
- develop implants, scaffolds, or modified surgical techniques to delay joint degeneration.
We pride ourselves in an ‘outside-the-box’ approach to understanding the mechano-biological response of the knee joint and to developing innovative approaches to the clinically relevant problem of knee joint degeneration after injury. Our models span the spectrum from cells, to tissues, to joints and as such allow for a comprehensive study of the effect of injury and repair on joint tissue response.
Patient-Specific Implants and Planning
The goal of Dr. Maher’s laboratory is to understand the relationship between knee joint contact mechanics and the biological response of articular cartilage and meniscus. This information is used to direct the development of implants, scaffolds, or modified surgical techniques to intervene in the degenerative process that occurs after injury. Her laboratory is currently developing biomaterials for joint restoration that can be used to treat young active patients with chondral or meniscal defects early in the course of the problem, thus delaying the need for a total joint replacement.

Joint-level loading systems: to apply physiological loads to mimic every-day activities
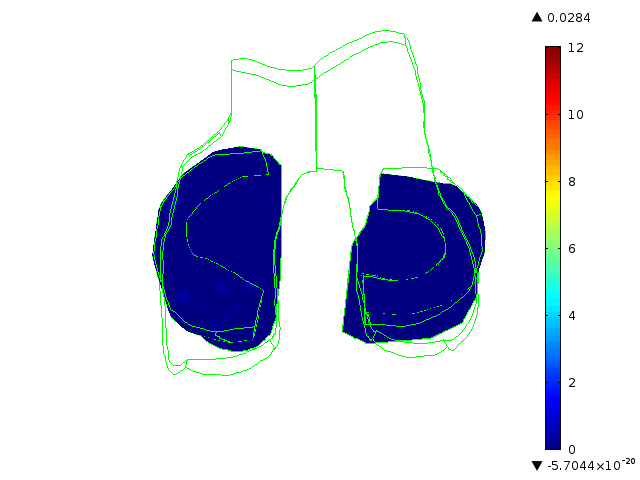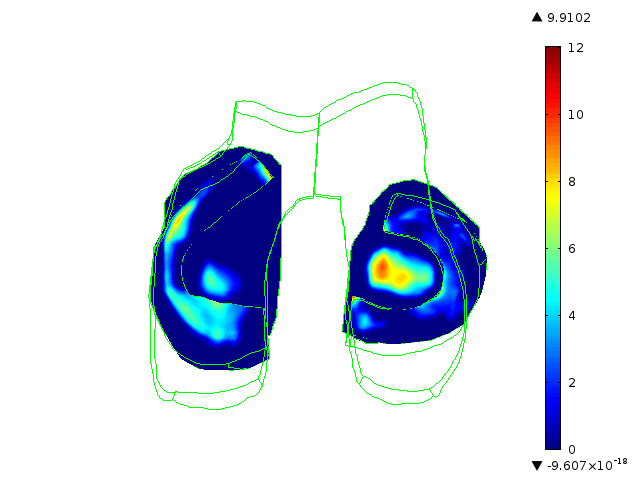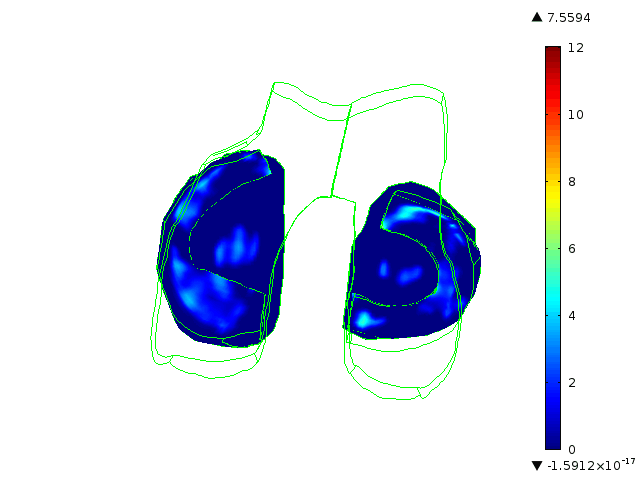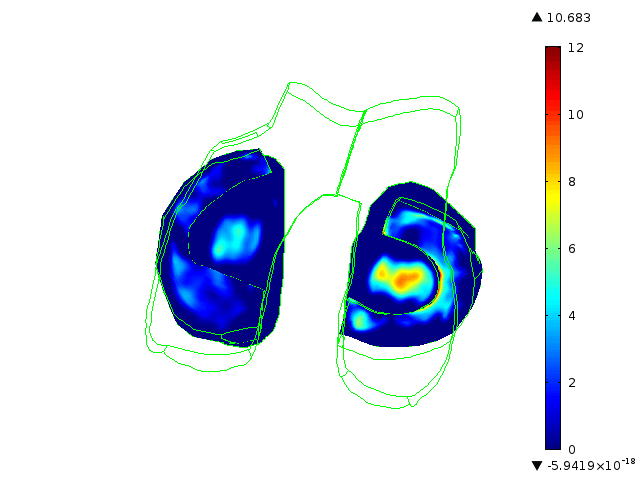
Statistically augmented computational models in which the effect of knee-specific variables can be parametrically analyzed
The Maher team has built multi-scale, multi-system models to help them understand how knee-specific factors affect joint contact mechanics and how injury affects the mechanics of the knee joint. They are using the models to assess the ability of scaffolds/ implants to restore pre-injury mechanics to the knee joint and to assess the ability of implants to integrate with the host tissue. One such implant for the treatment of cartilage defects is currently at an advanced level of pre-clinical testing, supported in part by the BioAccelerate NYC Program.
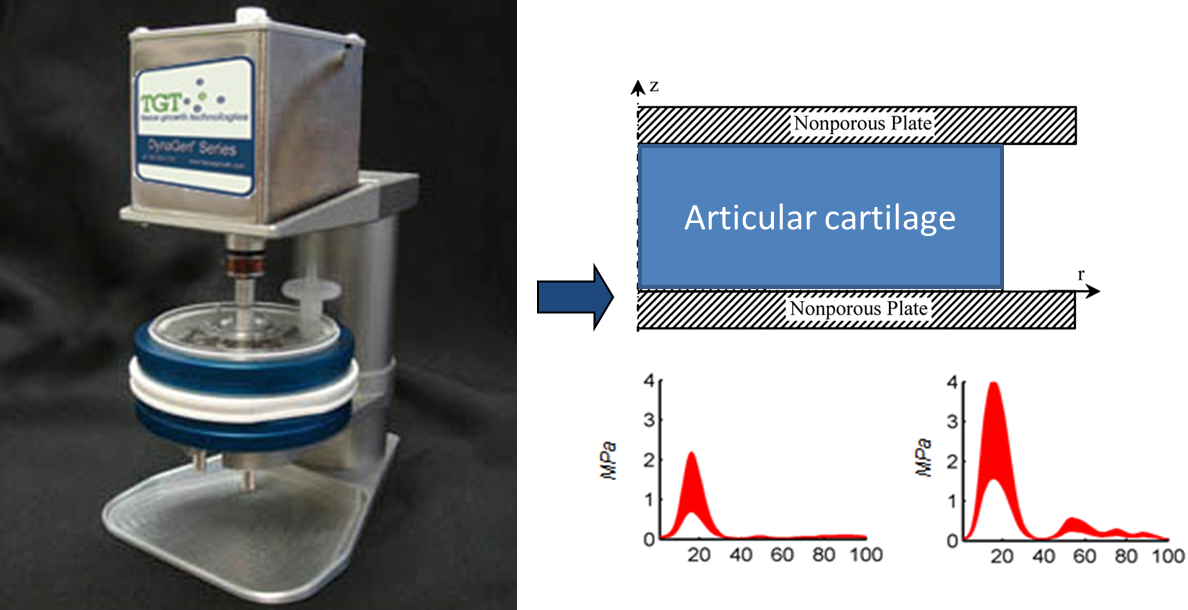
Bioreactors in which specific loading profiles can be applied to tissue explants, or scaffold-tissue constructs
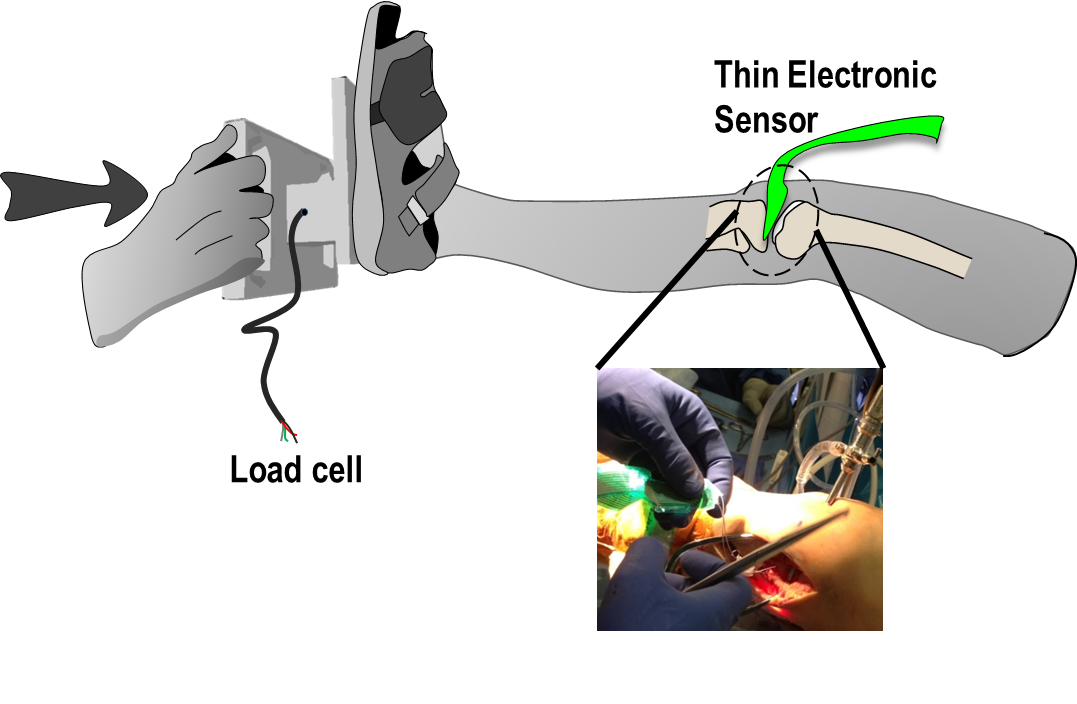
Patient-based models where contact mechanics can be measured during surgery
Laboratory Personnel
Our laboratory consists of mechanical engineers, biologists, chemical engineers and orthopaedic surgeons from the Sports Medicine Service. We have multidisciplinary collaborations with statisticians, and computational engineers which enable us to combine state-of-the-art cadaveric, computational, statistical, tissue explant and patient-based models.
Principal Investigator
Suzanne Maher, PhD, Associate Scientist & Laboratory Director
Assistant Scientist
Research Engineer
Research Technicians
OPPORTUNITIES TO JOIN OUR GROUP:
Research position available:
Post-doctoral fellowship in cadaveric simulator testing
HSS Collaborators
Russell Warren, MD
Peter Torzilli, PhD
Scott Rodeo, MD
Timothy Wright, PhD
Matt Koff, PhD
Hollis Potter, MD
External partnerships
Amy Lerner, PhD - University of Rochester
Tom Santner, PhD - The Ohio State University
Erin Leatherman, PhD - Kenyon College
Markus Wimmer, PhD - Rush University
Susan Chubinskaya, PhD - Rush University
Matthew Posner, PhD - United States Military Academy West Point
Alumni
Full Time:
Funmi Adebayo, PhD, Assistant Dean of Inclusion and Diversity, University of Massachusetts at Amherst, MA
Aliza Allon, PhD, McKinsey and Company
Devon Charlton, MD, Medical Resident, Danbury Hospital
Kirsty Culley, PhD, Science Engagement Manager at Anglia Innovation Partnership LLP
Patrick Donnelly, PhD, School of Dental Medicine, University of Pittsburgh
Jordan Green, University of Chicago Medical School
Hongqiang Guo, PhD, Senior Data Scientist at GNS Healthcare
Horng-Chaung Hsu, MD, Professor and Chairman Department of Orthopaedic Surgery, China Medical University Hospital, Taichung, Taiwan.
Kiwon Joh, Co-Founder at Blocklink Ventures
Mehdi Khoshgoftar, PhD, Sr. Computational Material Scientist at ASML and Adjunct Assistant Scientist at HSS
Cathal Moran, Professor of Orthopaedics and Sports Medicine; Consultant Orthopaedic Surgeon, Sports Surgery Clinic, Ireland
Kenneth W Ng, PhD, Assistant Medical Director for Health Science Communications
Lorenzo Pio Serino, Development Scientist, Orthox, UL
Alexander Shields, BS, Full time student at Duke University
Hongsheng Wang, PhD, Hardware Engineer, Google Wearable Technologies
Florian Wanivenhaus, Balgrist University Hospital, Department of Orthopaedics, University of Zurich, Switzerland
Supansa Yodmuang, PhD, Assistant Scientist, University of Thailand
Part Time:
John Anderson, MD, Rothman Institute Orthopaedics
Asheesh Bedi, MD, Ann Arbor, Michigan
Nicole Belkin, Columbia Orthopaedics
Robert Brophy, MD, Washington University Physicians
Saddiq El-Amin, MD, PhD, Southern Illinois University School of Medicine
Sommer Hammoud, MD, Rothman Institute Orthopaedics
Ian Hutchinson, Albany Medical College
Aaron Krych, MD, The Mayo Clinic
Moira McCarthy, MD, Hospital for Special Surgery
Michael Suk, Chairman, Department of Orthopaedic Surgery, Geisinger
Steven Thornton, MD, Texas Orthopaedic Associates
Dean Wang, MD, University of California, Irvine
Publications, Patents, and Funding Sources
Recent Publications
- Chen T, Brial C, McCarthy M, Warren RF, Maher SA. Synthetic PVA Osteochondral Implants for the Knee Joint: Mechanical Characteristics During Simulated Gait. Am J Sports Med. 2021 Sep;49(11):2933-2941
- Virk S, Meyers KN, Lafage V, Maher SA, Chen T. Analysis of the influence of species, intervertebral disc height and Pfirrmann classification on failure load of an injured disc using a novel disc herniation model. Spine J. 2021 Apr;21(4):698-707.
- Virk S, Chen T, Meyers KN, Lafage V, Schwab F, Maher SA. Comparison of biomechanical studies of disc repair devices based on a systematic review. Spine J. 2020 Aug;20(8):1344-1355.
- Maher SA, Kyle, D, Morrey B, Yazemsky M. Translating Orthopaedic Technologies into Clinical Practice: Challenges and Solutions J Am Acad Orthop Surg. 2019 Jan 1;27(1):e9-e16.
- Donnelly PR, Imbert L, Culley KL, Warren RF, Chen T, Maher SA. Self-Assembled Monolayers of Phosphonates Promote Primary Chondrocyte Adhesion to Silicon Dioxide and Polyvinyl Alcohol Materials, Journal of Biomaterials Science: Polymer Edition, Feb;30(3):215-232.
- Yodmuang S, Guo H, Brial C, Warren RF, Torzilli PA, Chen T, Maher SA. Effect of Interface Mechanical Discontinuities on Scaffold-Cartilage Integration. J Orthop Res. 2019 Apr;37(4):845-854. doi: 10.1002/jor.24238. Epub 2019 Mar 20.
- Brial C, McCarthy M, Chen T, Adebayo O, Wimmer M, Warren R, Maher SA. Lateral Meniscal Graft Transplantation: Effect of Fixation Method on Joint Contact Mechanics during Simulated Gait, American Journal of Sports Medicine, in press.
- Chen T, McCarthy MM, Guo H, Warren R, Maher SA. The Scaffold-Articular Cartilage Interface: A Combined In Vitro and In Silico Analysis Under Controlled Loading Conditions. J Biomech Eng. 2018 Sep 1;140(9).
- Guo H, Santner TJ, Lerner A, Maher SA. Reducing uncertainty when using knee-specific finite element models by assessing the effect of input parameters, Journal of Orthopaedic Research, 2017 Oct;35(10):2233-2242.
- Chen TO, Wang H, Warren R, Maher SA, Loss of ACL function leads to characteristic changes in tibial plateau dynamic contact stress profiles, Journal of Biomechanics, 2017 Aug 16;61:275-279.
- Donnelly PE, Chen T, Finch A, Brial C, Maher SA, Torzilli PA. Photocrosslinked tyramine-substituted hyaluronate hydrogels with tunable mechanical properties improve immediate tissue-hydrogel interfacial strength in articular cartilage. J Biomater Sci Polym Ed. 2017 Apr;28(6):582-600.
- Maher S, Wang H, Koff MF, Potter HG, Rodeo SA. A Clinical Platform for Understanding the Relationship Between Joint Contact Mechanics & Articular Cartilage Changes after Meniscal Surgery. Journal of Orthopaedic Research, 2017 Mar;35(3):600-611.
- Waldstein W, Perino G, Gilbert SL, Maher SA, Windhager R, Boettner F. OARSI osteoarthritis cartilage histopathology assessment system: A biomechanical evaluation in the human knee. J Orthop Res. 2016 Jan;34(1):135-40.
- Wang H, Koff MF, Potter HG, Warren RF, Rodeo SA, Maher SA. An MRI-compatible loading device to assess knee joint cartilage deformation: Effect of preloading and inter-test repeatability. J Biomech. 2015 Aug 13
- Donahue TL, Fisher MB, Maher SA. Meniscus mechanics and mechanobiology. J Biomech. 2015 Jun 1;48(8):1341-2.
- Wang H, Chen T, Gee AO, Hutchinson ID, Stoner K, Warren RF, Rodeo SA, Maher SA. Altered regional loading patterns on articular cartilage following meniscectomy are not fully restored by autograft meniscal transplantation. Osteoarthritis Cartilage. 2015 Mar;23(3):462-8.
- Guo H, Santner TJ, Chen T, Wang H, Brial C, Gilbert SL, Koff MF, Lerner AL, Maher SA. A statistically-augmented computational platform for evaluating meniscal function. J Biomech. 2015 Jun 1;48(8):1444-53. doi: 10.1016/j.jbiomech.2015.02.031. Epub 2015 Feb 26.
- Guo H, Maher SA, Torzilli PA. A biphasic finite element study on the role of the articular cartilage superficial zone in confined compression. J Biomech. 2015 Jan 2;48(1):166-70.
- Guo H, Maher SA, Torzilli PA. J Biomech. A biphasic multiscale study of the mechanical microenvironment of chondrocytes within articular cartilage under unconfined compression. 2014 Aug 22;47(11):2721-9.
- Leatherman ER, Guo H, Gilbert SL, Hutchinson ID, Maher SA, Santner TJ. Using a statistically calibrated biphasic finite element model of the human knee joint to identify robust designs for a meniscal substitute. J Biomech Eng. 2014 Jul 1;136(7).
- Guo H, Maher SA, Torzilli PA. A biphasic multiscale study of the mechanical microenvironment of chondrocytes within articular cartilage under unconfined compression. J Biomech. 2014 May 10
- Wang H, Gee AO, Hutchinson ID, Stoner K, Warren RF, Chen TO, Maher SA. Bone Plug Versus Suture-Only Fixation of Meniscal Grafts: Effect on Joint Contact Mechanics During Simulated Gait. Am J Sports Med. 2014 Apr 28. [Epub ahead of print]
- Wang H, Chen T, Koff MF, Hutchinson ID, Gilbert S, Choi D, Warren RF, Rodeo SA, Maher SA. Image based weighted center of proximity versus directly measured knee contact location during simulated gait. J Biomech. 2014 Apr 16
- McCarthy M, Maher SA, Bedi A, Warren R: Editorial, How can practitioners decide when to reconstruct a torn ACL? Clinical Practice, January 2014, Volume 11, Number 1.
- Wang H, Chen T, Torzilli P, Warren R, Maher S. Dynamic contact stress patterns on the tibial plateaus during simulated gait: a novel application of normalized cross correlation. J Biomech. 2014 Jan 22;47(2):568-74.
- Khoshgoftar M, Torzilli PA, Maher SA. Influence of the Pericellular and Extracellular Matrix Structural Properties on Chondrocyte Mechanics. J Orthop Res. 2017 Oct 16.Bedi A, Chen T, Santner TJ, El-Amin S, Kelly NH, Warren RF, Maher SA. Changes in dynamic medial tibiofemoral contact mechanics and kinematics after injury of the anterior cruciate ligament: a cadaveric model. Proc Inst Mech Eng H. 2013 Sep;227(9):1027-37.
Patents
- Interconnected porous non-degradable poly(vinyl) alcohol implant and method of manufacture, USPO: 8,440,618.
- Composition for the attachment of implants to collagen or other components of biological tissue, USPO 8,557,270.
- Device, method of implantation & instrumentation for the treatment of cartilage defects; USPTO 10,583,224.
- Instrumented boot for quantifying intra-operative joint contact mechanic; USPTO 10,070,972.
Funding Sources
- National Institutes of Health (NIAMS)
- NIH, NIAMS, AR075523: How Partial Meniscectomy Affects Contact Mechanics and Tissue Response. Dr. Scott Rodeo and Dr. Suzanne Maher, Co-PIs
- NIH, NIAMS, AR078751: Combined Engineering and Orthopaedics Training Program. Dr. Suzanne Maher and Dr. Marjolein van der Meulen, Co-PIs
- SBIR grant awarded to Hydro-Gen LLC (PI: Tony Chen, PhD); subcontract to Hospital for Special Surgery R43AR067533-01: Design Optimization of a non-degradable synthetic device for cartilage defects.
- 5T32 AR007281-30: Musculoskeletal Research Training Grant; S.A. Maher & M.C. van der Meulen
- R01 AR066635: Mechanobiological Risk Factors for Initiation of Post-Traumatic Osteoarthritis
- Widgeon Point Foundation
- The Russell Warren Chair in Tissue Engineering
- New York City Investment Fund & New York City Economic Development Corporation (BioAccelerate)
Back to HSS Research Institute