Research Manager
Neurology Research
The Department of Neurology at Hospital for Special Surgery (HSS) is at the forefront of advancing neurological science through clinical, translational, and basic research. Our nationally recognized investigators are dedicated to understanding and treating complex conditions, with specific focuses on neuromuscular diseases and movement disorders.
Our programs concentrate on exploring molecular mechanisms underlying skeletal muscle injury and repair, determining and controlling risk factors of neurological complications associated with orthopedic diseases and surgeries to improve surgical outcomes, pioneering innovative therapies for neuromuscular diseases such as amyotrophic lateral sclerosis, peripheral neuropathy, and myasthenia gravis, and identifying new biomarkers and therapeutic targets for movement disorders such as Parkinson's disease. At HSS Neurology, research is not only a pursuit of scientific inquiry but also a commitment to transforming patient care through discovery, collaboration, and translational excellence.
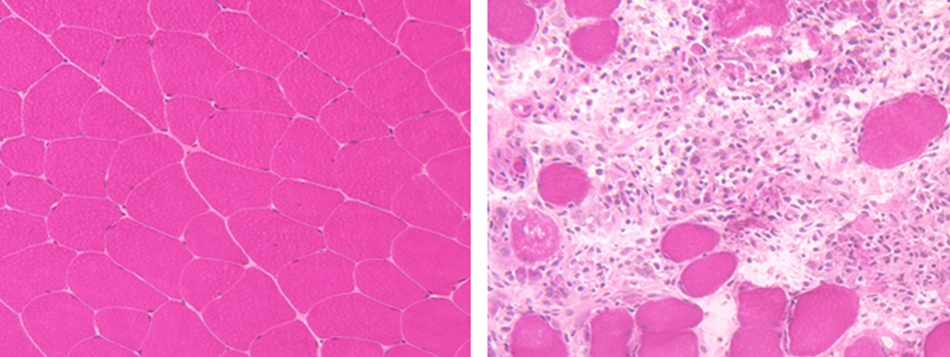
High-resolution microscopy showing the contrast between organized normal muscle (left) and the disrupted patterns of injured tissue (right).
Research Programs
Neuromuscular Research Program
This program integrates cutting-edge basic science with clinical application, supported by a dynamic portfolio of investigator-initiated studies and industry-sponsored clinical trials. These studies characterize neuromuscular complications associated with orthopedic diseases and surgeries, test innovative therapies for neuromuscular diseases such as amyotrophic lateral sclerosis, myasthenia gravis, and chronic inflammatory demyelinating polyneuropathy, explore molecular and cellular mechanisms underlying skeletal muscle injury and repair, and test novel therapeutic strategies to promote muscle injury repair. The program aims to enhance targeted interventions that improve patient outcomes via active research.
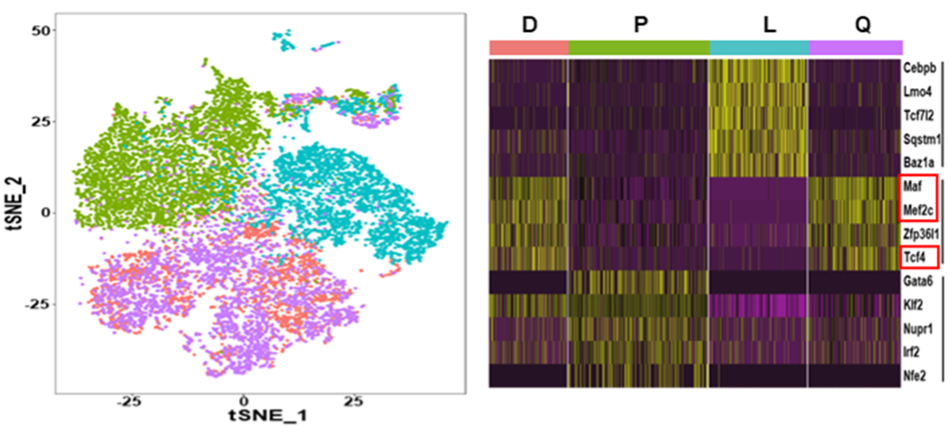
High-dimensional data visualization showing single-cell RNA sequencing clusters (left) and differential gene expression patterns (right) involved in skeletal muscle injury and repair.
Movement Disorder Research Program
This pioneering translational research initiative focuses on Parkinson’s disease and tremor. Led by a physician-scientist with formal training in molecular neuroscience and clinical neurology, the program bridges laboratory and bedside through ongoing research. The ongoing clinical trials & NIH funded research in the field of movement disorders contribute to the understanding of neurodegenerative mechanisms. The research is poised to identify new biomarkers and therapeutic targets, enhancing our understanding of the disease and opening doors to potential disease-modifying treatments.
At HSS, our research studies are the bridge between laboratory discovery and life-changing clinical care. We are currently conducting a range of industry-sponsored and investigator-initiated trials aimed at unlocking new treatments for complex neuromuscular and movement disorders. By participating in these studies, patients play a vital role in advancing the future of neurology.
Research Studies
Phase 3 CAPTIVATE Trial for Chronic Inflammatory Demyelinating Polyneuropathy (CIDP)
Sponsor: Dianthus
Site Principal Investigator: Dora Leung, MD
Site Co-Investigator: Erin Manning, MD
Contact: Kathleen Peterson
Tel: 917-260-4904
Email: neurologyresearch@hss.edu
HSS Neurology is participating in a new phase 3 clinical trial evaluating DNTH103, an investigational therapy for CIDP. Led by Principal Investigator Dr. Dora Leung, this trial marks a significant step forward in the development of targeted immunotherapies for chronic neuromuscular disease. DNTH103 is an extended half-life human IgG4 monoclonal antibody that selectively inhibits activated C1s, the initiating enzyme of the classical complement pathway. The study is designed in three parts: an open-label induction phase, a randomized double-blind placebo-controlled phase, and a long-term extension phase, collectively spanning over three years of clinical evaluation, including a 40-week safety follow-up. This trial not only investigates the therapeutic efficacy and safety of DNTH103, but also explores its potential to offer durable, precision-targeted immune modulation in CIDP. The study underscores HSS Neurology’s leadership in advancing mechanistic treatments in peripheral neuropathies through rigorously designed international clinical research.
Phase 3 Clinical Trial: Ulixcaltamide (PRAX-944) in Adults with Essential Tremor
Sponsor: Praxis Precision Medicine
Principal Investigator: Alexander Shtilbans, MD
This multicenter study, led by HSS neurologist Dr. Alexander Shtilbans, is to evaluate the therapeutic potential of an investigational drug to treat essential tremors in adults.
PROPEL — A New Phase 3, Multicenter, Randomized, Placebo-controlled, Double-blind Study to Assess the Efficacy and Safety of IMVT-1402 in Patients with Mild to Severe Generalized Myasthenia Gravis (gMG).
Site Principal Investigator: Dr. Dora Leung, MD
Site Co-Investigator: Erin Manning, MD
Contact: Kathleen Peterson
Tel: 917-260-4904
Email: neurologyresearch@hss.edu
HSS Neurology is participating in a Phase 3 global clinical trial evaluating IMVT-1402, a next-generation therapeutic monoclonal antibody for the treatment of gMG. Spearheaded at HSS by Dr. Dora Leung, this randomized, double-blind, placebo-controlled study investigates the efficacy, safety, and pharmacologic profile of IMVT-1402 in patients with mild to severe gMG. IMVT-1402 is a fully human anti-FcRn IgG1 monoclonal antibody, specifically engineered to reduce circulating pathogenic IgG autoantibodies, central drivers of gMG pathophysiology, while minimizing effector functions such as antibody-dependent cellular cytotoxicity. The study targets seropositive patients with anti-AChR, anti-MuSK, or anti-LRP4 antibodies and seeks to validate the role of FcRn inhibition as a precision approach in autoimmune neuromuscular disease. This trial represents a critical step in expanding treatment options for patients with refractory or difficult-to-manage gMG and reflects HSS Neurology’s leadership in advancing immunologically targeted therapies in complex autoimmune disorders. Through this trial, HSS Neurology contributes to expanding targeted, immunomodulatory treatment options in gMG, with the potential to improve functional outcomes and quality of life for patients living with this chronic autoimmune neuromuscular disorder.
MOBILIZE — A study Evaluating Efficacy and Safety of Riliprubart in Participants with Refractory Chronic Inflammatory Demyelinating Polyneuropathy (CIDP).
Site Principal Investigator: Dr. Dora Leung, MD
Site Co-Investigator: Erin Manning, MD
Contact: Kathleen Peterson
Tel: 917-260-4904
Email: neurologyresearch@hss.edu
This global, multicenter, placebo-controlled, Phase 3 study is designed to evaluate the efficacy and safety of riliprubart in adult participants with CIDP who have demonstrated insufficient response or refractoriness to standard-of-care therapies, including intravenous immunoglobulin, subcutaneous immunoglobulin, or corticosteroids. The study will follow a treatment duration of 48 weeks, with the primary objective being to assess riliprubart’s potential to improve clinical outcomes in this patient population. Participants must be at least 18 years of age at the time of signing the informed consent. This pivotal trial is poised to provide valuable insights into a promising therapeutic approach for CIDP, a rare and debilitating peripheral neuropathy, with the potential to significantly enhance treatment options for patients who have not responded adequately to conventional therapies.
VITALIZE — A Study Evaluating Efficacy and Safety of Riliprubart versus Intravenous Immunoglobulin (IVIg) in Participants with Chronic Inflammatory Demyelinating Polyneuropathy (CIDP).
Site Principal Investigator: Dr. Dora Leung, MD
Site Co-Investigator: Erin Manning, MD
Contact: Kathleen Peterson:
Tel: 917-260-4904
Email: neurologyresearch@hss.edu
This global, multicenter Phase 3 clinical trial investigates the efficacy and safety of riliprubart, an investigational complement pathway inhibitor, in adults with CIDP who continue to experience residual functional impairment despite maintenance IVIg therapy. The study is designed in two parts: an initial double-blind phase comparing riliprubart with IVIg, followed by an open-label extension in which all participants receive riliprubart monotherapy after IVIg withdrawal. By rigorously comparing these therapeutic strategies, this trial aims to advance the treatment landscape for CIDP and evaluate riliprubart’s potential to provide targeted, sustained disease control.
Data Registry for Neurological Complications Associated with Orthopedic Surgeries
Principal Investigator: Ashley Weng, MD
Co-Investigator: Lan Zhou, MD; PhD; Dora Leung, MD; Erin Manning, MD
Contact: Navdeep Singh Nayyar
Tel: 646-714-6135
Email: neurologyresearch@hss.edu
This Neuromuscular Registry at Hospital for Special Surgery is prospective research aiming at systematically capturing and analyzing neurological complications associated with orthopedic surgeries. The study will register all patients who require post-operative neurology consultation to study the incidence, risks, clinical presentations, diagnosis, management, and prognosis of neurological complications by analyzing perioperative variables, intraoperative neuromonitoring data, neuroimaging and electrodiagnostic test findings, and validated clinical outcomes. The findings may help guide prevention of complications and improvement of surgical outcomes.
Retrospective Study on the Effect of Corticosteroid Administration on Foot Drop following Total Knee Arthroplasty (TKA) and Total Hip Arthroplasty (THA)
Principal Investigator: Dr. Erin Manning, MD
Co-Investigator: Dora Leung, MD
Contact: Navdeep Singh Nayyar
Tel: 646-714-6135
Email: neurologyresearch@hss.edu
This retrospective study conducted at the Hospital for Special Surgery investigates the incidence, etiology, and recovery trajectories of postoperative foot drop following TKA) and THA. Key variables assessed include corticosteroid administration patterns, surgical and anesthetic factors, and detailed postoperative neurological outcomes. Data from diagnostic evaluations incorporating electromyography, nerve conduction studies, and advanced imaging modalities (MRI, CT) that were used to characterize the pattern and severity of neural impairment will be analyzed. This investigation provides critical insights into the pathophysiological underpinnings of postoperative foot drop and informs evolving perioperative strategies aimed at minimizing neurological complications while optimizing functional recovery in lower extremity arthroplasty patients.
Retrospective Study of Diffusion Tensor Imaging and Quantitative Susceptibility Mapping as Potential Biomarkers for Diagnosis and Prognostication in Patients with ALS and PLS
Principal Investigator: Dora Leung, MD
Co-Investigator: Erin Manning, MD
Contact: Navdeep Singh Nayyar
Tel: 646-714-6135
Email: neurologyresearch@hss.edu
This collaborative retrospective study between the Hospital for Special Surgery (HSS) and Weill Cornell Medicine (WCM) aims to evaluate advanced neuroimaging techniques, Diffusion Tensor Imaging (DTI) and Quantitative Susceptibility Mapping (QSM), as potential diagnostic and prognostic biomarkers in patients with Amyotrophic Lateral Sclerosis (ALS) and Primary Lateral Sclerosis (PLS). Given the clinical overlap and diagnostic complexity of these progressive motor neuron disorders, our goal is to determine whether quantitative imaging markers of white matter integrity and iron deposition correlate with disease severity and progression. By analyzing existing imaging and clinical data, this study seeks to enhance diagnostic accuracy, facilitate earlier intervention, and ultimately improve patient care. The findings may support the integration of DTI and QSM into routine MRI protocols, offering clinicians objective tools for differentiating ALS and PLS from mimicking neurological conditions.
Quantitative Mapping of Substantia Nigra Iron in Parkinson's Disease and Controls.
Principal Investigator: Alexander Shtilbans, MD
At the intersection of neurology, radiology, and neurosurgery, the study we are conducting explores the role of aberrant brain iron accumulation in the pathophysiology of Parkinson’s disease. Utilizing advanced magnetic resonance imaging techniques—specifically, quantitative susceptibility mapping (QSM), our research aims to non-invasively measure and characterize iron deposition in key deep brain nuclei across different stages of Parkinson’s disease. Preliminary findings demonstrate that iron accumulation is most pronounced in the substantia nigra and red nucleus, appearing early in the disease course and persisting as the condition progresses. Interestingly, other basal ganglia structures do not show similar alterations, suggesting a region-specific vulnerability. These insights not only advance our understanding of disease mechanisms but also highlight QSM's potential as a sensitive biomarker for disease staging and progression. The work reflects the commitment of scientists at Hospital for Special Surgery and Weill Cornell Medicine to pioneering diagnostic and translational approaches in neurodegenerative research.
Role of Macrophages in Skeletal Muscle Injury and Repair
Principal Investigator: Lan Zhou, MD; PhD
Co-Investigator: Xingyu Wang, PhD
Supported by NIH and institutions, Dr. Lan Zhou’s research team has been conducting this line of research for nearly 20 years. They have found that inflammation in response to acute skeletal muscle injury and chronic injury associated with muscular dystrophy is predominated by macrophage infiltration. Adequate infiltration of macrophages is essential to acute skeletal muscle injury repair, as macrophages phagocytose necrotic tissue and produce myotrophic growth factors to promote muscle regeneration. However, infiltrating macrophages contribute to muscle dystrophic changes in Duchenne muscular dystrophy. But blocking macrophage infiltration via CCR2 ablation is not sufficient to improve muscle dystrophy, because muscle resident macrophages expand and undergo pathogenic activation to contribute to muscle fibrosis. Currently, Dr. Zhou’s team is exploring whether inhibition of both inflammatory macrophage infiltration and resident macrophage expansion could represent a viable therapeutic approach to improve muscle pathology and function in Duchenne muscular dystrophy. The goal of this research is to explore a novel and useful macrophage-based therapy.
Role of Fibro/Adipogenic Progenitors in Skeletal Muscle Injury and Repair
Principal Investigators: Lan Zhou, MD; PhD and Xingyu Wang, PhD
This study is to dissect diverse roles of fibro/adipogenic progenitors (FAPs) in acute and chronic skeletal muscle injury and repair. Being the muscle mesenchymal stromal cells, FAPs play a critical role in promoting muscle injury repair or fibrosis via production of extracellular matrix proteins. Studies by Dr. Lan Zhou’s team and others, using single-cell RNA sequencing, demonstrate that FAPs have diverse functional subsets. They interact with other cells in injured muscles, influencing inflammation, regeneration, and injury repair. Dr. Zhou’s team is currently dissecting the interactions between FAPs and macrophages to further determine inflammation regulation by FAPs during skeletal muscle injury and repair.
Molecular Mechanisms of Paraspinal Muscle Fatty Degeneration Associated with Spine Scoliosis
Principal Investigators: Sohrab Virk, MD; Lan Zhou, MD; PhD and Xingyu Wang, PhD
Collaborating with spine surgery team, this study will explore molecular pathogenic mechanisms underlying paraspinal muscle atrophy and fatty infiltration associated with degenerative spine scoliosis. The goal of this research is to develop targeted interventions to reduce paraspinal muscle degeneration, promote muscle regeneration and function, and improve outcomes before and after spine surgery.
Characterization of Muscle Fatty Degeneration Associated with Massive Rotator Cuff Tear
Principal Investigators: Claire Eliasberg, MD and Xingyu Wang, PhD
Co-Investigators: Lan Zhou, MD; PhD and Scott Rodeo, MD
Rotator cuff tears are the most common cause of pain and disability related to the shoulder. About 25% of rotator cuff repairs fail to heal, and the presence of rotator cuff muscle atrophy and fatty degeneration is a known independent risk factor. Over one-third of patients with full-thickness rotator cuff tears have fatty degeneration of supraspinatus or infraspinatus muscles. Collaborating with physician-scientists in orthopedic surgery, this study will characterize the timing and nature of muscle pathological changes arising after massive rotator cuff tears, as well as the underlying molecular and cellular mechanisms. The goal of this study is to develop novel treatments aimed at preventing or ameliorating muscle degenerative changes to improve patient outcomes.
Principal Investigators
Clinical Research Support Team
-
-
Research Coordinator
Basic Science Research Team
-
Professor
-
Assistant Scientist
Publications
- Wang X and Zhou L. (2026) Macrophage Infiltration, Activation, and Therapeutic Implication in Skeletal Muscle Injury and Repair. Int J Mol Sci. 27(3):1332. doi: 10.3390/ijms27031332. PMID: 41683757
- Wang X, Moy JK, Wang Y, Smith GR, Ruf-Zamojski F, Przytycki PF, Sealfon SC, Zhou L. (2025) Heterogeneous Macrophage Activation in Acute Skeletal Muscle Sterile Injury and mdx5cv Model of Muscular Dystrophy. Int J Mol Sci 26 (16):8098. doi: 10.3390/ijms26168098. PMID: 40869418
- Wang Y, Wang X, Alabdullatif S, Homma ST, Alekseyev YO, and Zhou L. (2025) Expansion and pathogenic activation of skeletal muscle resident macrophages in mdx5cv/Ccr2-/- mice. Proc Natl Acad Sci U S A. 122 (11) e2410095122 https://doi.org/10.1073/pnas.2410095122
- Burkhard MD, Evangelisti G, Altorfer FCS, Paschal PK, Achebe CC, Gorgy G, Kelly MJ, Zelenty WD, Girardi FP, Lebl DR, Hughes AP, Cammisa FP, Sama AA, Emerson RG, Sokunbi G. Is Intraoperative Neuromonitoring with SSEPs and EMG Predictable for Postoperative Neurologic Deficit in Posterior Lumbar Fusion Surgery? A Retrospective Cohort Analysis.Global Spine J. 2025 May 8:21925682251341820. doi: 10.1177/21925682251341820. Online ahead of print. PMID: 40340552
- Tanwar M, Miller E, Tsiouris AJ, Nguyen J, Agarwal V, Marinelli L, Erdemir GA, Shetty T. White Matter Hyperintensities on High-Resolution 3-T MRI: Frequency in Mild Traumatic Brain Injury and Associations With Clinical Markers-A Prospective Controlled Multicenter Study.AJR Am J Roentgenol. 2025 May;224(5): e2432274. doi: 10.2214/AJR.24.32274. Epub 2025 Feb 19. PMID: 39969142
- Shtilbans A, Esneault E, Simon F, Mazzulli JR, Quiriconi DJ, Rom D, Reintsch WE, Krahn AI, Durcan TM. Evaluation of Additive Neuroprotective Effect of Combination Therapy for Parkinson's Disease Using In Vitro Models.Antioxidants (Basel). 2025 Mar 27;14(4):396. doi: 10.3390/antiox14040396. PMID: 40298667
- Zarr VM, Liou JY, Merricks EM, Davis TS, Thomson K, Greger B, House PA, Emerson RG, Goodman RR, McKhann GM 2nd, Sheth SA, Schevon CA, Rolston JD, Smith EH. Protocol for detecting and analyzing non-oscillatory traveling waves from high-spatiotemporal-resolution human electrophysiological recordings.STAR Protoc. 2025 Mar 21;6(1):103659. doi: 10.1016/j.xpro.2025.103659. Epub 2025 Feb 27. PMID: 40022738
- Wang Y, Wang X, Alabdullatif S, Homma ST, Alekseyev YO, Zhou L. Proc Natl Acad Sci U S A. 2025 Mar 18;122(11):e2410095122. doi: 10.1073/pnas.2410095122. Epub 2025 Mar 11. PMID: 40067893
- Homma ST, Wang X, Frere JJ, Gower AC, Zhou J, Lim JK, tenOever BR, Zhou L. (2024) Respiratory SARS-CoV-2 Infection Causes Skeletal Muscle Atrophy and Long-Lasting Energy Metabolism Suppression. Biomedicines 12(7), 1443; https://doi.org/10.3390/biomedicines12071443
- Shtilbans A, Reintsch WE, Piscopo VEC, Krahn AI, Durcan TM. Combination of tauroursodeoxycholic acid, co-enzyme Q10 and creatine demonstrates additive neuroprotective effects in in-vitromodels of Parkinson's disease.Front Neurosci. 2024 Dec 23;18:1492028. doi: 10.3389/fnins.2024.1492028. eCollection 2024. PMID: 39764390
- Kelly MJ, Burkhard MD, Altorfer FCS, Emerson RG, Sama AA. Intradural Disk Herniation at the Conus Medullaris: A Case Report with Emphasis on Patient Positioning and Neuromonitoring.JBJS Case Connect. 2024 Nov 22;14(4):e24.00235. doi: 10.2106/JBJS.CC.24.00235. eCollection 2024 Oct 1. PMID: 39841040
- Lamothe MA, Erickson J, Emami K.Management of Concussion in the Young Athlete.HSS J. 2024 Aug;20(3):402-408. doi: 10.1177/15563316241238014. Epub 2024 Mar 18. PMID: 39108449
- Rucker S, Singh N, Mai E, Asada T, Shahi P, Mercado K, Leung D, Iyer S, Emerson R, Qureshi SA. Feasibility of Saphenous Nerve Somatosensory-Evoked Potential Intraoperative Monitoring During Lumbar Spine Surgery: Early Results.Spine (Phila Pa 1976). 2024 Jul 1;49(13):923-932. doi: 10.1097/BRS.0000000000004938. Epub 2024 Jan 26. PMID: 38273786
- Manning E, Emerson R. Intraoperative Monitoring of Scoliosis Surgery in Young Patients.J Clin Neurophysiol. 2024 Feb 1;41(2):138-147. doi: 10.1097/WNP.0000000000001058. PMID: 38306222
- Wang X, Zhou L. The multifaceted role of macrophages in homeostatic and injured skeletal muscle. Front Immunol. 2023 Oct 25;14:1274816. doi: 10.3389/fimmu.2023.1274816. PMID: 37954602; PMCID: PMC10634307.
- Abrams RMC, Zhou L, Shin SC. (2023) Persistent post-COVID-19 neuromuscular symptoms. Muscle Nerve. 68(4):350-355.
- Wang X, Chen J, Homma S, Wang Y, Smith GR, Ruf-Zamojski F, Sealfon SC, Zhou L. (2023) Diverse Effector and Regulatory Functions of Fibro/Adipogenic Progenitors during Skeletal Muscle Fibrosis in Muscular Dystrophy. iScience. PMID: 36594034 DOI: 10.1016/j.isci.2022.105775.
- Scholtz LC, Rosenberg J, Robbins MS, Wong T, Mints G, Kaplan A, Leung D, Kamel H, Ch'ang JH. Ultrasonography in neurology: A comprehensive analysis and review.J Neuroimaging. 2023 Jul-Aug;33(4):511-520. doi: 10.1111/jon.13124. Epub 2023 May 19. PMID: 37204265
- Kim N, Jamison K, Jaywant A, Garetti J, Blunt E, RoyChoudhury A, Butler T, Dams-O'Connor K, Khedr S, Chen CC, Shetty T, Winchell R, Hill NJ, Schiff ND, Kuceyeski A, Shah SA. Comparisons of electrophysiological markers of impaired executive attention after traumatic brain injury and in healthy aging. 2023 Jul 1;274:120126. doi: 10.1016/j.neuroimage.2023.120126. Epub 2023 Apr 30. PMID: 37191655
- Wang X, Zhou L. (2022) The Many Roles of Macrophages in Skeletal Muscle Injury and Repair. Front Cell Dev Biol. 10:952249.
- Abrams RMC, Simpson DM, Navis A, Jette N, Zhou L., Shin SC. (2022) Small fiber neuropathy associated with SARS-CoV-2 infection. Muscle Nerve. 65(4):440-443.
- Marmor WA, McElheny KD, Lingor RJ, Shetty T, Lin E, Casey E, Ling DI. Disparities in concussion diagnoses compared to ankle injuries.PM R. 2023 Mar;15(3):325-330. doi: 10.1002/pmrj.12793. Epub 2022 Apr 8. PMID: 35191195
- Zhou L. (2021) Small fiber neuropathy in the elderly. Clin Geriatr Med. 37(2):279-288.
- Zhou L., Siao P. (2021) Lateral femoral cutaneous neuropathy caused by prone positioning to treat COVID-19-associated acute respiratory distress syndrome. Muscle Nerve. 63: E50-E52.
- Li KR, Avecillas-Chasin J, Nguyen TD, Gillen KM, Dimov A, Chang E, Skudin C, Kopell BH, Wang Y, Shtilbans A. Quantitative evaluation of brain iron accumulation in different stages of Parkinson's disease.J Neuroimaging. 2022 Mar;32(2):363-371. doi: 10.1111/jon.12957. Epub 2021 Dec 14. PMID: 34904328
- Smith EH, Liou JY, Merricks EM, Davis T, Thomson K, Greger B, House P, Emerson RG, Goodman R, McKhann GM, Sheth S, Schevon C, Rolston JD. Human interictal epileptiform discharges are bidirectional traveling waves echoing ictal discharges.Elife. 2022 Jan 20;11:e73541. doi: 10.7554/eLife.73541. PMID: 35050851
- Wang X, Sathe AA, Smith GR, Ruf-Zamojski F, Nair V, Lavine KJ, Xing C, Sealfon SC, Zhou L. (2020) Heterogeneous origins and functions of mouse skeletal muscle-resident macrophages. Proc Natl Acad Sci U S A. 117(34):20729-20740.
- Rubenstein AB, Smith GR, Raue U, Begue G, Minchev K, Ruf-Zamojski F, Nair VD, Wang X, Zhou L., Zaslavsky E, Trappe TA, Trappe S, Sealfon SC (2020). Single-cell transcriptional profiles in human skeletal muscle. Sci Rep. 10(1):229. doi: 10.1038/s41598-019-57110-6.
- Merricks EM, Smith EH, Emerson RG, Bateman LM, McKhann GM, Goodman RR, Sheth SA, Greger B, House PA, Trevelyan AJ, Schevon CA. Neuronal Firing and Waveform Alterations through Ictal Recruitment in Humans.J Neurosci. 2021 Jan 27;41(4):766-779. doi: 10.1523/JNEUROSCI.0417-20.2020. Epub 2020 Nov 23. PMID: 33229500
- Garvey MD, Miller CJ, Kim EU, Skulikidis G, Shetty T. Post-concussion Vulnerability to Transient Global Amnesia.Front Neurol. 2020 Nov 12;11:517863. doi: 10.3389/fneur.2020.517863. eCollection 2020. PMID: 33262736
- Smith EH, Merricks EM, Liou JY, Casadei C, Melloni L, Thesen T, Friedman DJ, Doyle WK, Emerson RG, Goodman RR, McKhann GM 2nd, Sheth SA, Rolston JD, Schevon CA. Dual mechanisms of ictal high frequency oscillations in human rhythmic onset seizures.Sci Rep. 2020 Nov 5;10(1):19166. doi: 10.1038/s41598-020-76138-7. PMID: 33154490
- Reisener MJ, Shue J, Hughes AP, Sama AA, Emerson RG, Guheen C, Beckman JD, Soffin EM. Hemodynamically significant cardiac arrhythmias during general anesthesia for spine surgery: A case series and literature review.N Am SpineSoc J. 2020 Jun 12;2:100010. doi: 10.1016/j.xnsj.2020.100010. eCollection 2020 Aug. PMID: 35141581
HSS Neurology News
Research Grant
R01 research grant supporting collaborative work in synucleinopathies
Principal Investigator: Jacqueline Burré, PhD; Weill Cornell Medicine
Co-Investigator: Alexander Shtilbans, MD PhD; Hospital for Special Surgery
HSS Neurology is proud to announce its participation in a newly awarded NIH R01 research grant supporting collaborative work in synucleinopathies. As part of this initiative, Alexander Shtilbans, MD, PhD is contributing to the project titled “Changes in Synaptic Vesicle-Binding of Alpha-Synuclein as an Early Biomarker for Synucleinopathies” (R01NS136423), in partnership with Jacqueline Burré, Ph.D. from Weill Cornell Medicine. This translational study aims to identify early molecular biomarkers of Parkinson’s disease and dementia with Lewy bodies by examining alterations in alpha-synuclein vesicle-binding in both the brain and enteric nervous system. By leveraging clinical recruitment, deep phenotyping, and human gut biopsies collected during routine colonoscopies, the project seeks to establish a prodromal biomarker platform to guide early diagnosis and intervention in neurodegenerative disease.
Meeting Presentation
HSS Neurology will present the following studies in the 2026 American Academy of Neurology Annual Meeting
- Weight Drop and Foot Drop: A Case Series of GLP-1 Agonist-Associated Peroneal Neuropathy
Gautham Updrasta, Lan Zhou, Eerin Manning, Dora Leung, MD
Department of Neurology
Hospital for Special Surgery - Steroids May Not be Helpful for Recovery in Foot Drop after TKA and THA: A Retrospective Cohort Study
Erin Manning MD, Kathleen Peterson BS, Navdeep S. Nayyar MD
Department of Neurology
Hospital for Special Surgery - Combined Efficacy and Safety of Ulixacaltamide in Essential Tremor: Topline Results from the Phase 3 Essential3 Program
Alexander Shtilbans1,2*, Jill Farmer3*, Salima Brillman4*, Monique Giroux5, Melanie Fridley5, Kimberly Crimin5, Minao Tang5, Taylor Crush5, Claudio Santos5, Steven Petrou5, Alyssa Wyant5*, Megan Sniecinski5*, Marcio Souza5*, on behalf of the Essential3 Study Team
1Hospital for Special Surgery, Department of Neurology, New York, NY, USA
2Weill Cornell Medicine, Department of Neurology, New York, NY, USA
3BoroNeuro, Hopewell, NJ, USA
4Parkinson's Disease and Movement Disorders Center of Silicon Valley, Palo Alto, CA, USA
5Praxis Precision Medicines, Boston, MA, USA - Amitriptyline for Acute Mild Traumatic Brain Injury (mTBI)
Ava Panetto and Teena Shetty
Department of Neurology, Hospital for Special Surgery
Research Highlights








